Diapharm is a market leader in the development and marketing authorisation of herbal medicinal products, no matter whether it involves products with well-established use (WEU), regular marketing authorisations or simplified traditional registrations. More than 500 herbal medicinal products in Europe are based on dossiers prepared by Diapharm. Marketing authorisation holders value our professional expertise and many years of experience in terms of marketing authorisations, registrations and new developments.
Many plants have a long history of use as part of self-medication. In some cases, knowledge of the medicinal properties of valerian, hawthorn, St John’s wort, arnica and other plants has been passed down from one generation to the next for centuries.
Diapharm provides traders of herbal medicinal products with a comprehensive range of phytopharmaceutical portfolios that cover a broad spectrum of indications. How does this benefit you? You can quickly position your company in the market with a strong brand – and there are no long lead times, because Diapharm has already taken care of the required approvals and registrations.
Our clients in this field include many of Europe’s best-known herbal medicinal companies. They come to Diapharm to place a full portfolio of herbal medicinal products in the market on short notice – with the assistance of Diapharm’s own herbal registrations and licenses, and supported by our licensing service for projects and dossiers.
Diapharm leads the market in developing and registering traditional herbal medicinal products – commonly known as traditional herbal registrations or THRs. More than 500 herbal medicinal products in Europe are based on dossiers prepared by Diapharm.
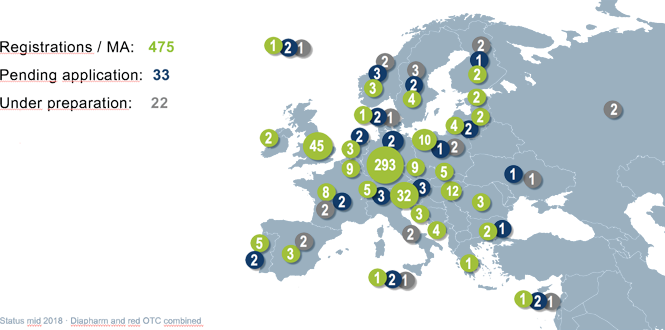
We have already prepared complete CTD dossiers for herbal medicinal products. These are ready to be used in registration procedures for traditional herbal medicinal products (THMP) including evidence of traditional use as well as for Well-Established Use (WEU) medicines.
To ensure that traditional herbal medicinal products can retain a long-term place in the modern market for OTC medicinal products, the EU has established a simplified “traditional herbal registration” (THR) for them: the registration process as per EU directive 2004/24/EC which meanwhile has been transferred to national law in all member states.
This ensures that the efficacy of traditional herbal medicinal products (THMPs) is considered plausible without the need for conducting extensive clinical studies. Instead, the applicant must prove that the product has been in medicinal use for at least 30 years, with at least 15 of them in Europe.
The THMP indication approved by the regulatory authority reads: “A traditional herbal medicinal product […] exclusively based upon long-standing use.”
Monographs from the Herbal Medicinal Products Committee (HMPC) of the EMA, London, are available almost solely for mono products. As a result, applicants need to provide their own documents to prove the efficacy of combination products. The analysis of such combinations is often significantly more challenging. Diapharm offers a wide range of pre-formulated mono and combination products with innovative indications. From a marketing perspective, combinations are usually far more interesting.
Diapharm is also active in the field of traditional Chinese medicine (TCM). We focus on helping pharmaceutical companies from China transfer their herbal TCM products to the European system of traditional registration (EU Directive 2004/24/EC) and gain market access.